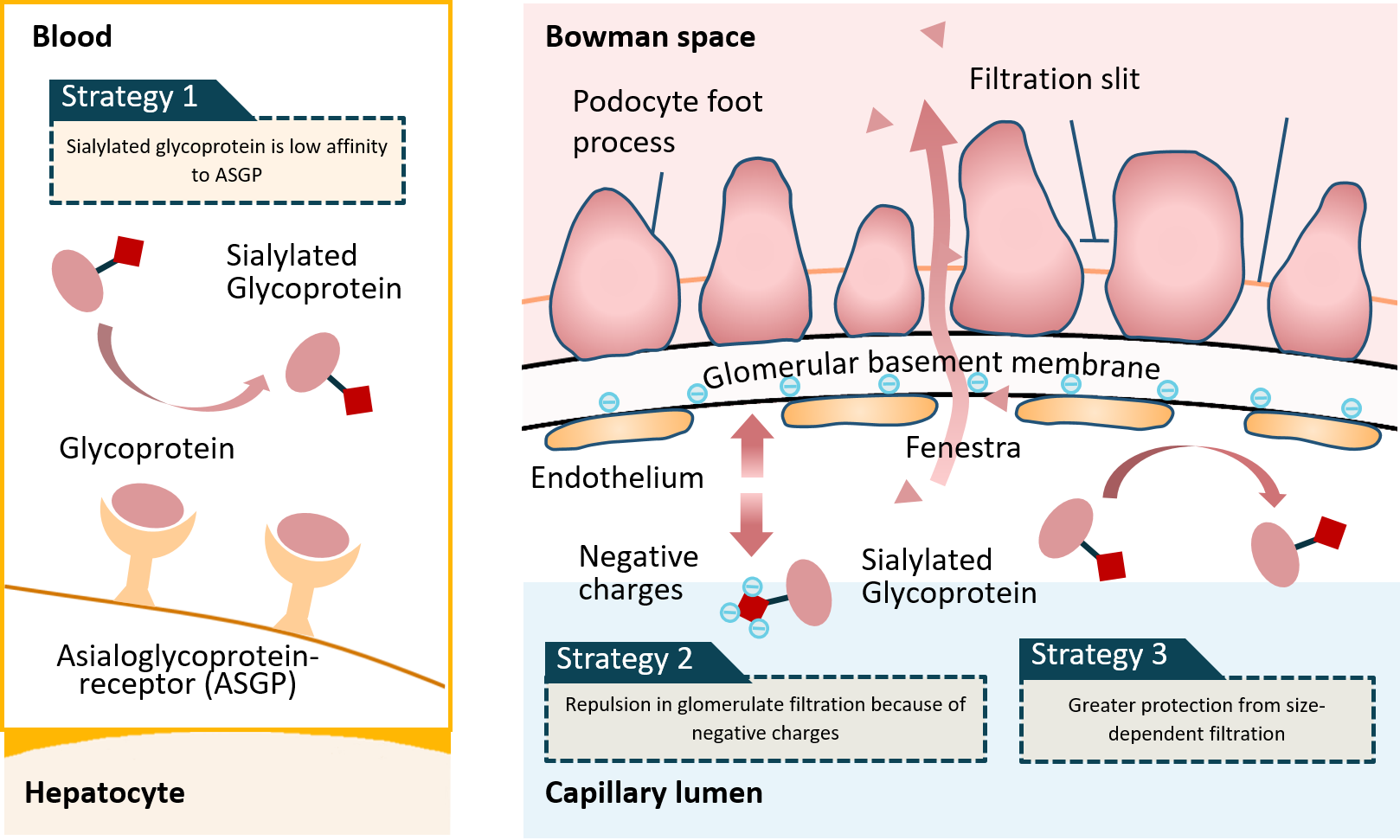
Sialic acids are critical factors determining the half-life of glycoproteins in circulation. There is a positive correlation between half-life and sialic acid content in glycoproteins (e.g. Erythropoietin). The proteins clearance occurs mainly at the liver and kidneys. The sialyated glycoprotein has a low affinity to asialoglycoprotein-receptor (ASGP) on the hepatocyte. In addition, the negatively charged sialyated glycoproteins exert a repulsive force in glomerulate filtration of kidney. It also exerts a greater protection from size-dependent filtration in kidney.
We have employed this mechanism to develop our next-generation long acting protein drugs by employing our “Proteoglycan Fusion Protein Platform” to design proteins carrying a higher content of sialic acids which has an extended half-life, enhanced biological activity, and of low risk in the anti-drug antibody induction thus with low safety concerns of liver/kidney toxicity.
sFc Fusion Protein Platform
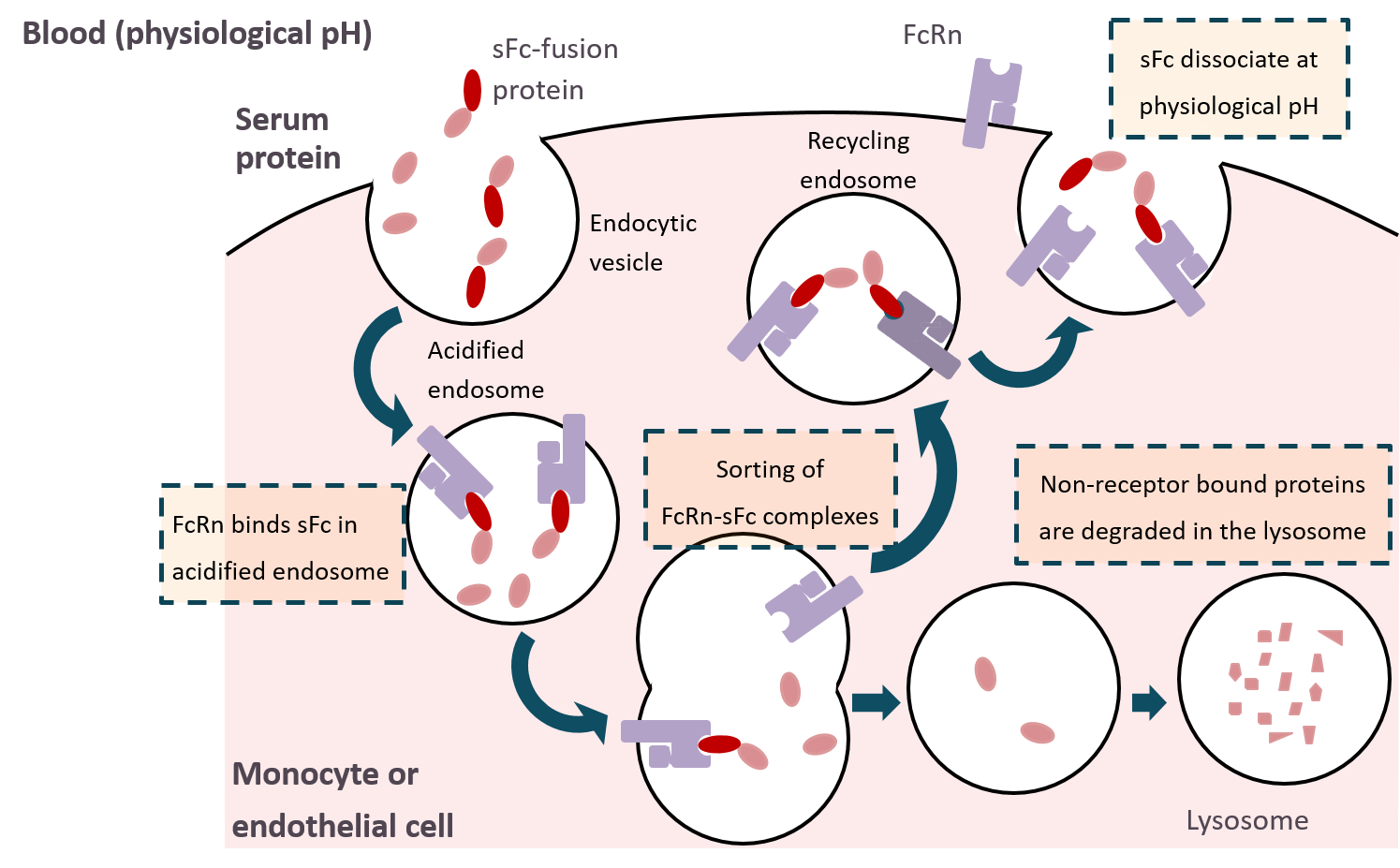
IgG is secreted as a monomer that is small in size to allow easy perfusion into tissues. The IgG neonatal Fc receptor (FcRn) is normally expressed in human placenta and epithelial cells and participates in an endocytic salvage pathway that prevents degradation of IgG. The high affinity of IgG for FcRn at acidic pH is to result in binding of internalized IgG to FcRn after uptake into acidic endosomes. Most soluble proteins are directed to lysosomes after internalization. Internalized FcRn-bound IgG returns to the plasma membrane and is effectively rescued from the default degradative pathway. Upon exposure to the neutral pH of the extracellular space, IgG can then dissociate from FcRn and return to circulation. Thus, the extended serum half-life property of the antibody is retained in the Fc fragment. This salvage pathway provides another mechanism for development of next-generation protein drugs that have a prolonged half-life in blood circulation, thus allowing for less frequent dosing over an extended treatment period.
Our proprietary “Single Chain Fc (sFc) Platform” is to use our innovative “Single Chain Fc (sFc) Platform” to create fusion proteins will be with extended half-life and enhanced biological activity accompanied by an easy manufacturing process.
Microsphere Technology Platform
One major challenge for successful administration of peptide and protein drugs is to maintain their stability and effective concentration during delivery. Among the various commonly adopted means, micrometer-size polymer carriers, such as PLGA microspheres, offer popular drug delivery technology. In addition to the tissue compatibility and biodegradability, PLGA microspheres are capable of releasing the encapsulated drugs slowly in the human body while maintaining their effective concentration, thereby lowering administration frequency. The drug-releasing duration can even be adjusted by modifying the composition and side chains of the polymer. PLGA microspheres are usually fabricated for use as injectable, and their production requires high-level technique and manufacturing processes.
We are developing long-acting injectables by applying this microsphere technology platform to a peptide drug for hormone and cancer therapy with formulations which offer a stable and zero-order controlled-release delivery over a one-month or longer period.
